The Puzzle Most of Us Never Solve
We have all been there. We apply the recommended dose of fertilizer. We water on time. We use good quality seeds. And yet — the crop looks pale, growth is slow, and yields disappoint. We blame the weather. We blame the seed company. We increase the fertilizer dose next season, hoping more will fix it.
But the real culprit is quietly sitting in our soil — and it has nothing to do with how much fertilizer we apply. It has everything to do with whether our soil’s pH is allowing our crops to absorb what we are giving them.
This one number — soil pH — could be the reason everything else we do on our farm either works or fails.
So What Is Soil pH?
pH is a measure of how acidic or alkaline our soil is. It runs on a scale of 0 to 14. A pH of 7 is neutral. Anything below 7 is acidic. Anything above 7 is alkaline.
Most of our crops — wheat, rice, maize, vegetables, pulses — grow best in a pH range of 6.0 to 7.5. This is the sweet spot where nutrients are most available, soil organisms are most active, and roots can absorb what they need without resistance.
The moment our soil moves outside this range — even by half a point — the consequences are surprisingly serious.
The Nutrient Lock — Why pH Matters More Than Fertilizer Dose
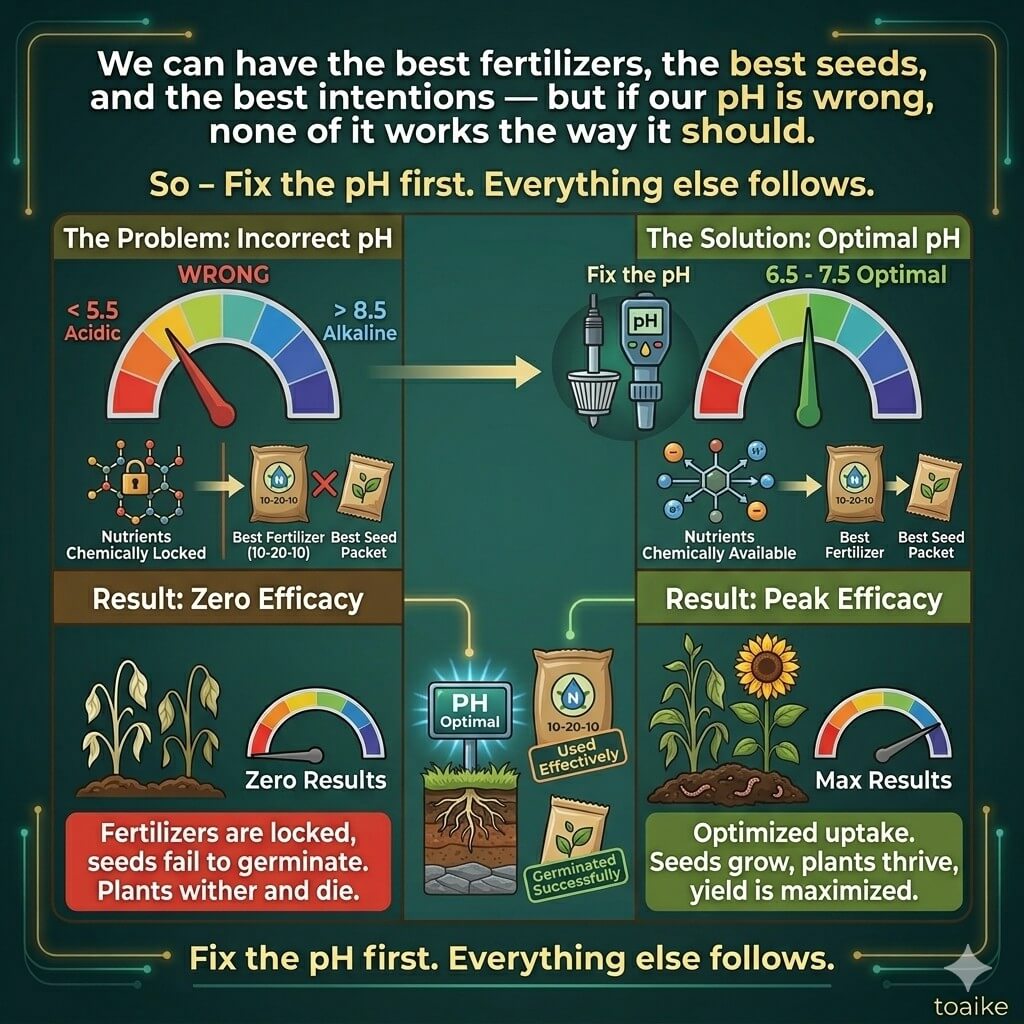
Here is the fact that changes everything: nutrients do not become available to plants simply because they are present in the soil. They become available only when the pH is right.
Think of soil pH as a key. Our fertilizers are the food we are trying to deliver to our crops. But if the pH is wrong, the door stays locked — no matter how much food we pile up outside it.
In acidic soils (pH below 6.0):
- Nitrogen becomes less available as microbial activity slows down
- Phosphorus gets locked up, binding tightly to iron and aluminium compounds
- Toxic levels of aluminium and manganese become active — directly damaging roots
- Calcium and Magnesium deficiencies become common, weakening plant structure
In alkaline soils (pH above 7.5):
- Phosphorus again becomes unavailable — this time binding to calcium
- Micronutrients like Zinc, Iron, Boron, and Manganese get locked out
- Zinc deficiency — already the most widespread micronutrient problem in Indian soils — becomes acute
- Soil structure often deteriorates, leading to crusting and poor water infiltration
This is precisely why we can spend thousands of rupees on fertilizers and see little to no response — if our pH is off, our investment simply does not reach the crop.
What Causes Soil pH to Change?
Soil pH is not fixed. It shifts over time — sometimes naturally, often because of what we do on our land. Understanding what drives these changes helps us stay ahead of the problem before it affects our crops.
Overuse of Nitrogen Fertilizers — The Biggest Culprit Every time we apply urea or ammonium-based fertilizers in excess, we are quietly acidifying our soil. As these fertilizers break down, they release hydrogen ions into the soil — and it is hydrogen ions that drive pH downward. Season after season of heavy nitrogen application without organic matter to buffer it is one of the leading causes of soil acidification on intensively farmed land. We often do not notice it happening — until our yields start telling us something is wrong.
Continuous Cropping Without Rotation Plants absorb alkaline nutrients — calcium, magnesium, and potassium — from the soil as they grow. When we grow the same crop repeatedly without rotation or replenishment, these nutrients are steadily removed and never replaced. Over time, their absence tips the soil toward acidity. Crop rotation and legume inclusion naturally slow this process down.
Heavy Rainfall and Leaching In high-rainfall regions, water continuously moves through the soil, carrying alkaline minerals like calcium and magnesium downward and out of the root zone. What remains behind is a more acidic soil profile. This is a natural process — but it is accelerated when soil organic matter is low and there is nothing to hold those minerals in place.
Irrigation Water Quality The water we use for irrigation carries its own chemistry. Alkaline or hard water — high in calcium carbonate and bicarbonates — gradually raises soil pH with every irrigation cycle. This is a common cause of creeping alkalinity in canal-irrigated and groundwater-irrigated fields, particularly in dry regions where evaporation leaves mineral deposits behind in the soil.
Organic Matter Depletion Organic matter acts as a natural pH buffer — it resists dramatic swings in either direction. When we reduce the return of crop residues, stop applying manure, and rely entirely on chemical inputs, we strip the soil of this buffering capacity. Without it, pH becomes increasingly unstable and vulnerable to the acidifying or alkalising effects of everything else we apply.
Industrial and Urban Pollution Acid rain — formed when industrial emissions of sulphur dioxide and nitrogen oxides dissolve in rainwater — deposits acidic compounds directly onto our farmland. In areas near factories, brick kilns, or heavily trafficked roads, this is an ongoing and often overlooked contributor to soil acidification.
Natural Soil Parent Material Some soils are simply born acidic or alkaline — depending on the rock and mineral deposits from which they formed over thousands of years. Soils derived from granite or sandstone tend toward acidity. Those formed from limestone or calcium-rich deposits tend toward alkalinity. While we cannot change what our soil started as, we can manage what it becomes.
At a Glance — What Causes Soil pH to Change?

A quick way to read this table:
- If most of our farming practices fall in the first three rows — we are likely heading toward acidity
- If our irrigation water is hard or alkaline — we are likely heading toward alkalinity
- If our organic matter is low — our pH is unstable and vulnerable to all of the above
How Do We Fix It?
The good news is that pH correction is straightforward — provided we test first and act on what we find.
For Acidic Soils: Agricultural lime (calcium carbonate) or dolomite lime is the most widely recommended treatment for acidic soils. It gradually neutralises excess acidity and restores the pH to a range where nutrients become available again. Because lime works slowly, we should apply it at least one full season before planting. Adding organic matter alongside lime accelerates recovery. The quantity we need depends on how acidic our soil is and what type of soil we have — so we should always apply lime based on our soil test report or the recommendations on our Soil Health Card.
For Alkaline and Sodic Soils: Gypsum (calcium sulphate) is the most effective treatment for sodic soils — replacing the excess sodium that causes structural breakdown. Elemental sulphur helps lower pH over time in alkaline soils. The application rate varies significantly depending on the severity of the problem and our soil type — so we should follow the specific recommendation from our soil test report.
For Both: Regularly adding compost, Farm Yard Manure, and green manures buffers soil pH naturally — making it less prone to swinging toward extremes. Organic matter is the great stabiliser of soil chemistry.
Finally - The Simple Step We Keep Skipping
Before we plan our next crop, before we order fertilizers, before we prepare our field — we should test our soil pH. It takes minutes. It costs very little. And the information it gives us shapes every other decision we make that season.
Our Soil Health Card already carries our field’s pH reading along with specific correction recommendations. If we have not read that number yet — or if our card is more than two years old — now is the time to check.
Because here is the truth we cannot afford to ignore: we can have the best fertilizers, the best seeds, and the best intentions — but if our pH is wrong, none of it works the way it should.
So – Fix the pH first. Everything else follows.
Pingback: Potassium Solubilizing Bacteria KSB in Agriculture
Pingback: What is Fertilizer Burn