What is Salt Index in Fertilizers
Every fertilizer, when it dissolves in soil water, changes the chemistry of that water. Some change it a little; others change it dramatically. The salt index is the number that tells you exactly how dramatic that change is — and by extension, how carefully a particular fertilizer needs to be handled.
Formally, the salt index expresses the degree to which a fertilizer elevates the osmotic concentration of soil solution relative to a fixed standard. In everyday language: it is a score that reveals how much a fertilizer disrupts the soil’s water balance. A low score means gentle disruption. A high score means the soil chemistry is being pushed hard — and plants feel that push.
Simple version: The salt index tells a farmer how ‘aggressive’ a fertilizer is in the soil. The higher the score, the more carefully it must be dosed and watered in.
How The Salt Index is calculated
The measurement method was established in 1943 by researchers Rader, White, and Whittaker and has remained the benchmark ever since. The reference compound is sodium nitrate, which is assigned a fixed value of 100. All other fertilizers are then tested under controlled conditions and scored proportionally against that standard.
What is actually being measured is the rise in osmotic pressure per unit weight of fertilizer applied. Two factors drive this rise:
- The number of ions produced — a fertilizer that splits into many particles when dissolved creates higher osmotic pressure than one that releases fewer. More particles in solution means more pressure on plant roots.
- The solubility of the compound — highly soluble fertilizers dissolve rapidly and release all their ions quickly, creating an immediate spike in osmotic pressure. Slow-dissolving or coated products release ions gradually, keeping pressure manageable.
A fertilizer scoring 75 — like urea — pushes osmotic pressure to 75% of what sodium nitrate would produce. A fertilizer scoring 116 — like Muriate of Potash — exceeds the benchmark entirely, making it the most osmotically aggressive common fertilizer available in the Indian market.
The Science in Plain Language
When any fertilizer granule makes contact with soil moisture, it breaks apart into its constituent ions. Urea, for example, splits through hydrolysis into ammonium and carbonate ions. DAP releases ammonium and phosphate. MOP releases potassium and chloride. Each of these dissolved particles adds to the total concentration of the soil solution.
Plant roots absorb water through a process called osmosis — water moves from an area of lower concentration, inside the soil pores, into the higher concentration environment of the root cells. This is the normal, healthy direction of flow. When fertilizer is applied in excess, the dissolved ions raise the concentration of soil water above that of the root cells, and osmosis reverses. Water begins moving out of the roots and into the soil rather than inward, leaving the plant dehydrated from within.
The analogy: imagine trying to drink a glass of very salty water. Instead of hydrating you, the salt draws moisture from your body to dilute itself. Excess fertilizer does exactly this to a plant’s root system.
The salt index predicts how easily this reversal can occur. Fertilizers with a high index can tip the balance with a relatively small overdose; those with a low index have a much wider margin of safety before any harm results.
Salt Index of Some fertilizers
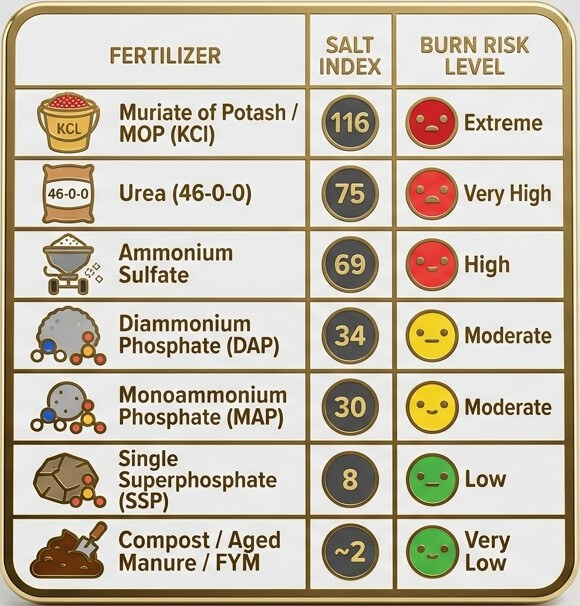
Note: Salt index values are measured per equal weight of fertilizer. Since different fertilizers are applied at different rates per acre, always consider total dose alongside the index — not the index alone.
Why Salt Index Matters To a Farmer
India’s farming conditions make salt index awareness particularly important. Large portions of the country’s cultivated land face one or more of the following challenges that heighten fertilizer burn risk:
- Light, sandy soils — common across Rajasthan, parts of Maharashtra, and coastal regions — hold far less water than heavier soils, meaning dissolved salts concentrate faster and at greater intensity around roots.
- Long dry periods — applying high-index fertilizers like urea or MOP during summer or drought spells, when soil moisture is already low, dramatically increases osmotic stress on crops.
- Reliance on urea — India’s heavily subsidised urea programme has made urea the default nitrogen input for millions of farmers. Its salt index of 75 means it demands respect — correct dosing, split application, and thorough irrigation after each application.
- Stacking multiple chemical inputs — applying urea, DAP, and MOP together in a single basal dose is common practice but stacks three different salt contributions simultaneously, compounding total osmotic pressure on the root zone.
Understanding salt index helps a farmer make smarter choices at every stage — what to buy, how much to apply, when to apply it, and how much water to follow it with.
Three Rules Every Farmer Should Follow
Rule 1 — The index is a guide, not a verdict
A high salt index fertilizer applied at the correct rate, on moist soil, with adequate follow-up irrigation is generally safe. The index tells you the potential for harm — the dose, timing, and water management determine whether that potential is realised. Urea at the recommended rate, watered in well, feeds a crop. Urea double-dosed on dry ground in May burns it.
Rule 2 — Soil type changes everything
The same salt index fertilizer behaves differently depending on what it lands in. Clay soils buffer osmotic changes because they hold more water; light sandy soils do not. A dose perfectly safe on a well-irrigated black cotton soil can cause significant burn on a shallow laterite or sandy loam. Always adjust rates downward on light soils and upward irrigation accordingly.
Rule 3 — Organic inputs occupy a different category entirely
Farmyard manure, vermicompost, neem cake, oilseed cakes, and green manures all have salt indexes close to zero. This is not because they contain less nitrogen — it is because their nitrogen is bound inside organic molecules that soil microbes must decompose before any ion is released into solution. There is no sudden osmotic spike. This is the fundamental reason why soils managed with organic inputs are far more forgiving and resilient than those relying entirely on chemical fertilizers.
At The End
Salt index = how hard a fertilizer pushes on your soil’s water balance. Know the number, respect the dose, follow with water — and your crops will never know the difference between a bag of fertilizer and a good meal.